A team of researchers, led by Professor Yong-Tae Kim and Ph.D. candidate Sang-Hoon You at POSTECH, has made a significant breakthrough in the development of fuel cell catalysts for hydrogen-powered cars. Their study, published in ACS Energy Letters, focuses on combating corrosion in fuel cells by tailoring the hydrogen oxidation reaction to match the hydrogen concentration within the cell.
Fuel cells used in automobiles are prone to degradation, particularly in the cathode catalyst, which is regularly exposed to start-up and shut-down events. This is especially relevant for automotive fuel cells that frequently undergo cycles of start-up and shut-down.
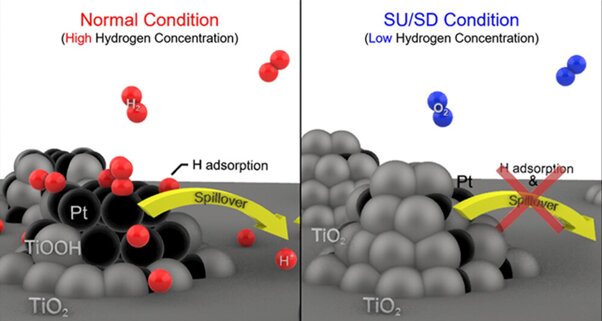
During normal operation, fuel cells receive a steady supply of high-concentration hydrogen. However, when the car is turned off or started, the hydrogen concentration temporarily decreases, leading to the unintentional triggering of an oxygen reduction reaction in the anode when external air mixes with the remaining hydrogen. This, in turn, causes sudden potential jumps and carbon corrosion in the cathode.
To address this issue, the research team developed a catalyst called Pt/TiO2, consisting of platinum (Pt) deposited onto titanium dioxide (TiO2). This catalyst effectively prevents corrosion in fuel cells used in hydrogen-powered cars. The catalyst’s performance is derived from the strong interaction between titanium dioxide and platinum, as well as the ability of hydrogen spillover to modify the surface conductivity of the catalyst based on the hydrogen concentration.
When a vehicle starts or stops suddenly, the hydrogen concentration within the fuel cell decreases accordingly. As a result, the titanium dioxide expands onto the platinum, burying it beneath the catalyst’s surface. This burying of platinum, caused by the expansion of titanium dioxide, transforms the catalyst into an insulator due to the low conductivity of titanium dioxide. This insulating effect hampers the catalyst’s ability to conduct electricity, preventing unwanted oxygen reduction and potential jumps in the cathode.
On the other hand, during normal vehicle operation with high hydrogen concentration, the conductive platinum is exposed on the catalyst’s surface, and titanium dioxide reduction occurs. This promotes hydrogen mobility on the catalyst’s surface through a phenomenon called hydrogen spillover, enhancing current flow and facilitating the hydrogen oxidation reaction.
To compare the newly developed catalyst with conventional ones, the research team conducted simulation tests. The results demonstrated that fuel cells using the Pt/TiO2 catalyst exhibited three times higher durability compared to traditional fuel cells. This signifies a successful increase in fuel cell durability through the selective oxygen reduction and hydrogen oxidation reactions based on the hydrogen concentration.
If this research can address the existing durability challenges faced by fuel cells in hydrogen-powered vehicles, it has the potential to enhance the status of Korean hydrogen-fueled automobiles in the next-generation mobility industry.
Source: Pohang University of Science and Technology