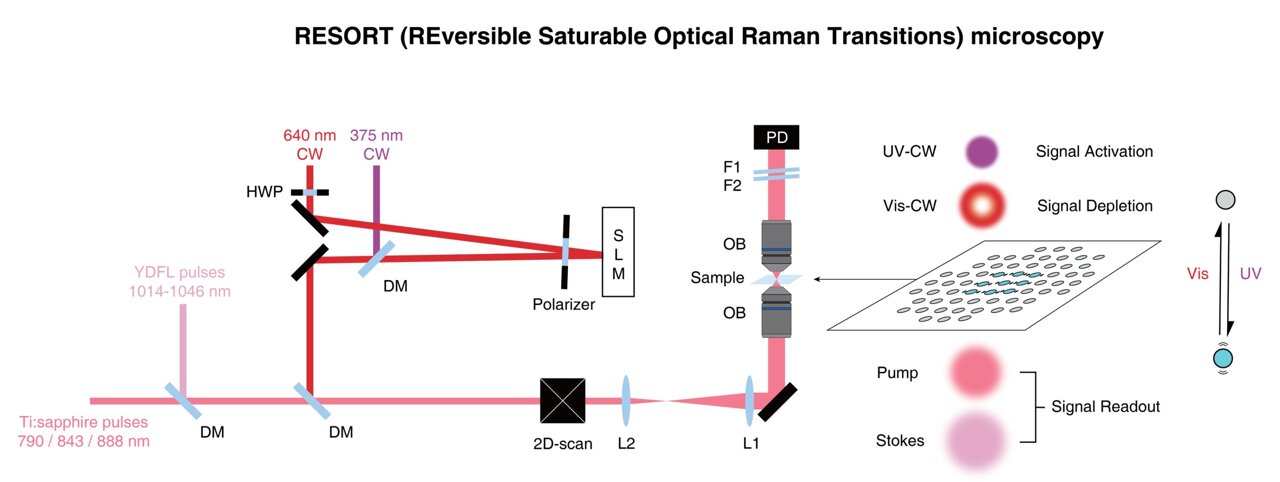
New imaging technique combines the benefits of super-resolution fluorescence and vibrational imaging
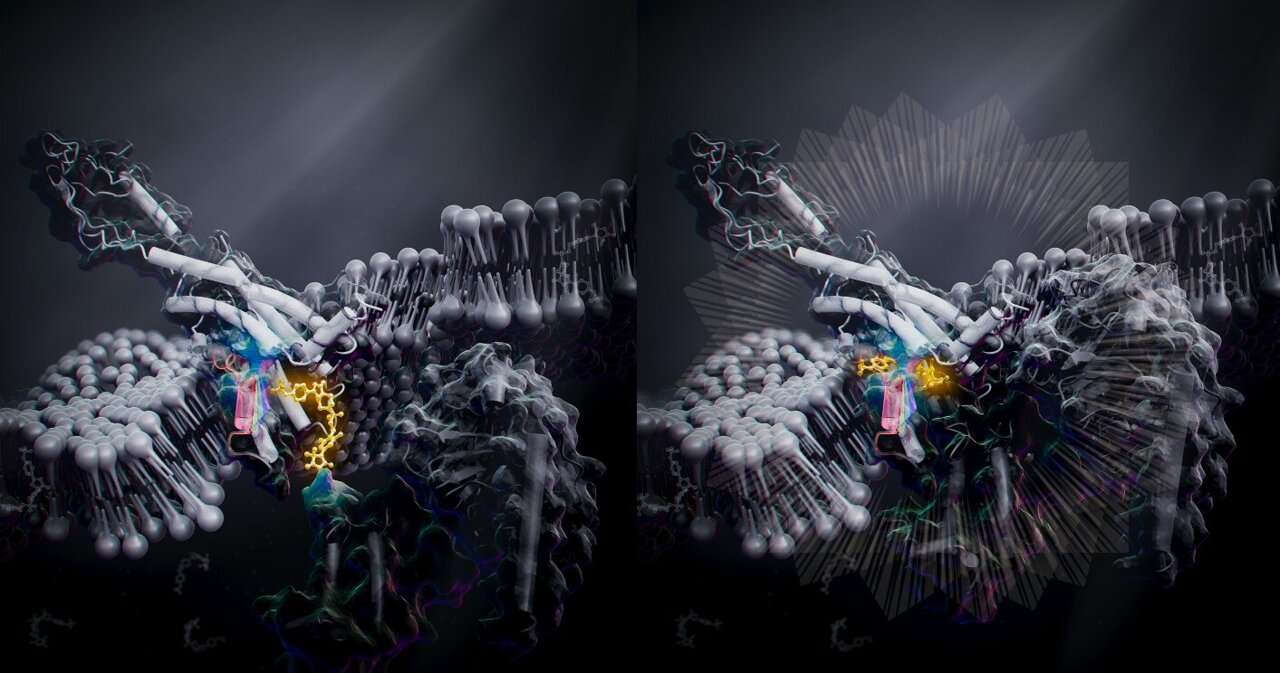
Scientists from the University of Tokyo, in collaboration with other researchers, have developed a groundbreaking imaging method called RESORT that allows for unprecedented observation of living systems. Published in the journal Science Advances, this innovative approach combines features from two leading imaging techniques to analyze biological samples in greater detail. Throughout history, humans have utilized … Read more