New research conducted at the Perelman School of Medicine at the University of Pennsylvania is shedding light on the reasons why some individuals are more prone to allergies than others. The study focused on the role of a protein called ETS1 and how genetic variations affecting this protein can impact our body’s response to allergies.
The researchers discovered that even small alterations in ETS1 can increase the likelihood of allergic reactions that lead to inflammation. The findings of this study were recently published in the journal Immunity.
According to the United States Centers for Disease Control and Prevention, allergies rank as the sixth most prevalent cause of chronic illness in the country, resulting in an annual expenditure exceeding $18 billion. While previous research has established a strong genetic basis for allergies and identified specific genetic variations associated with these chronic diseases, the mechanisms by which our DNA influences our susceptibility to allergies have remained unclear. However, understanding this relationship could pave the way for improved research and potential new treatments.
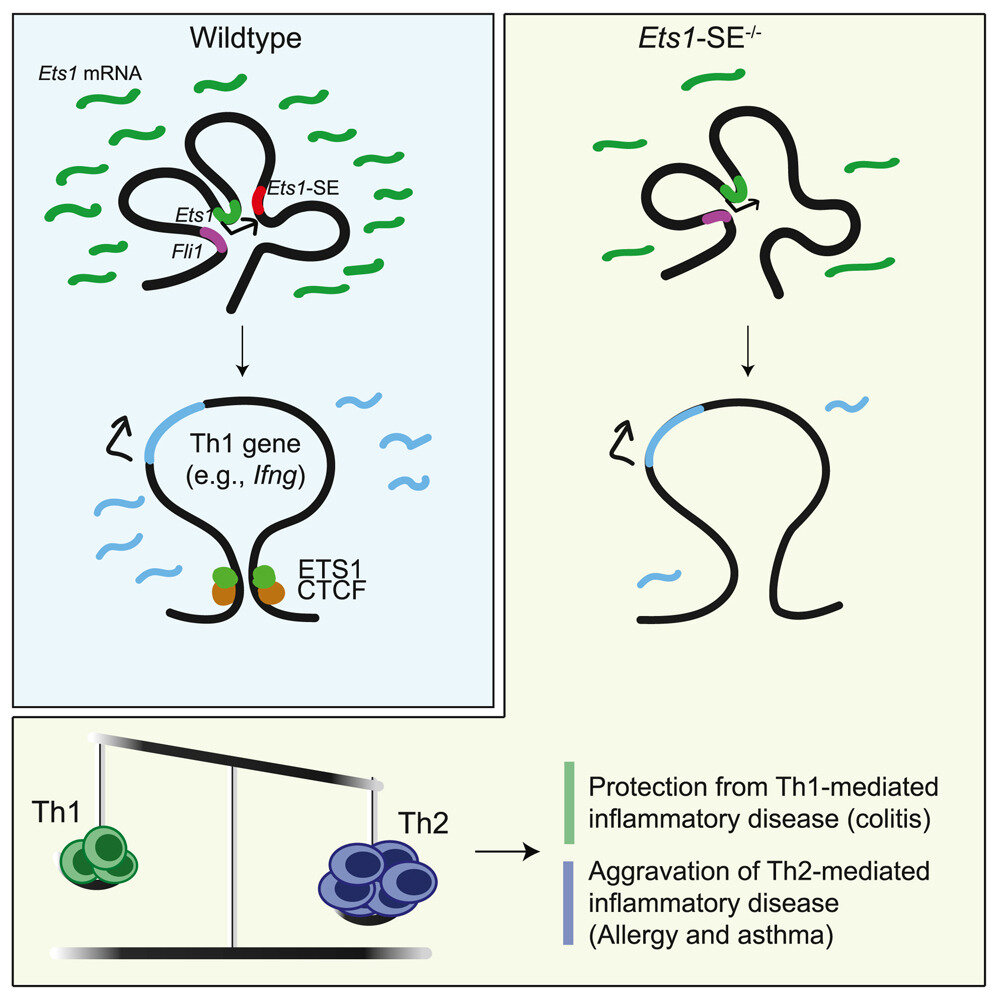
Using advanced genomics and imaging techniques, a collaborative team of researchers, led by Dr. Golnaz Vahedi, an associate professor of Genetics, and Dr. Jorge Henao-Mejia, an associate professor of Pathology and Laboratory Medicine, found that the ETS1 protein is involved in regulating a type of immune cell called CD4+ T helper cells. These cells play a crucial role in allergic reactions and coordinate the immune response by activating and coordinating other immune cells.
The interactions between DNA within the genomic segment encompassing the ETS1 gene control the production of the ETS1 protein. The researchers discovered that these interactions function similarly to a dimmer switch, where changes in the DNA sequence can disrupt the switch’s control over the ETS1 protein. This disruption can result in imbalances in immune cells and lead to allergic inflammation.
While significant progress has been made in understanding genetic traits that follow predictable patterns, such as those inherited from parents, it has been more challenging to comprehend conditions influenced by multiple genes and common in populations. These complex conditions cannot be attributed to a single “on/off” switch in a gene.
Instead, they may arise from subtle changes in DNA that affect the interactions between genes. However, researchers still have limited knowledge about how these DNA changes relate to gene organization and gene expression in most complex diseases.
“This study demonstrates how minor differences in our DNA can disrupt the equilibrium between our immune cells, resulting in observable characteristics in patients. This phenomenon may also occur in other common diseases, such as autoimmune disorders,” said Dr. Henao-Mejia.
Source: Perelman School of Medicine at the University of Pennsylvania